
Welcome to The Wellness Ledger
A weekly- health led newsletter grounded in evidence based medicine along with prospective randomized controlled trials by medical specialists. Our goal is to help you make sense of complex scientific information and turn it into clear, evidenced based practices you can use to make better decisions about your health and wellness.
The FDA just removed the black box warning from both estrogen and testosterone therapy.
Millions of women avoided hormone replacement therapy after a single 2002 headline told them it caused cancer and heart attacks. Their doctors agreed. The prescriptions stopped. What nobody said loudly enough: the study behind that headline was run on 63-year-olds who had been without estrogen for over a decade. Its findings were applied to 50-year-olds going through menopause. That was the part of the headline that was suddenly misleading.
In early 2025, the FDA removed the cardiovascular black box warning from all testosterone products for men. In November 2025, it removed the black box warnings from most HRT products for women. Two separate decisions. The same pattern: outdated fear, corrected by evidence.
This issue breaks down what the WHI (Women’s Health Initiative) actually says, what newer research reveals about who truly benefits from HRT, and exactly what you should ask at your next appointment.
TL;DR -- THE THREE THINGS THAT MATTER MOST
The WHI tested 63-year-olds. The results were applied to 50-year-olds. Mean age at enrollment: 63. Average years since menopause: 12. The findings were then universally applied to newly menopausal women. That mismatch is where 23 years of clinical fear began.
A 26% breast cancer risk increase is not what it sounds like. That relative figure translates to 8 extra cases per 10,000 women per year. Not 26 in every 100. Eight in ten thousand. The relative number made every headline. The absolute number did not.
In November 2025, the FDA removed the black box warning from most HRT products. After comprehensive reviews of 20+ years of evidence, including expert panel hearings in July 2025, the agency formally revised its stance. The research moved years ago. The label finally caught up.
WHAT CHANGED: THE FDA DECISION
The label that caused the collapse has been removed.
What the FDA removed in November 2025:
Black box warnings for cardiovascular disease, breast cancer, and probable dementia from most HRT products.
The 'lowest effective dose for shortest time' directive.
What the FDA added:
Affirmative guidance to consider initiating HRT in women under 60 or within 10 years of menopause onset, based on a favorable benefit-risk profile.
What did not change:
Black box warning for endometrial cancer on estrogen-alone products for women with an intact uterus. HRT is not approved solely for dementia or cardiovascular prevention to this point, although some studies suggest beneficial outcomes.
Source: HHS Fact Sheet, November 2025, hhs.gov
ALSO IN 2025: TESTOSTERONE
Most people heard about the estrogen decision. Fewer heard about the testosterone one.
In February 2025, the FDA removed the cardiovascular black box warning from all testosterone replacement therapy (TRT) products for men. The warning, added in 2014, said testosterone increased the risk of heart attacks and stroke. The evidence that prompted it came from two observational studies that were later heavily criticised for design flaws.
The TRAVERSE trial settled it. 5,246 men with hypogonadism and existing or high cardiovascular risk were randomised to testosterone or placebo for a median of 33 months. Cardiovascular event rate: 7.0% in the testosterone group versus 7.3% in placebo. Hazard ratio was deemed 0.96 (probability of a negative event occurring), which signifies a lower risk. The trial was powered for noninferiority and hit it cleanly.
A secondary finding from the T4DM trial (Wittert et al., Lancet Diabetes Endocrinol 2021): testosterone treatment for 2 years reduced new-onset type 2 diabetes by 22.5% (p = 0.029) in men with impaired glucose tolerance.
What the FDA did add: a new warning about blood pressure. TRT can potentially produce a measurable increase in blood pressure across product types, confirmed in post-market ambulatory monitoring studies. Routine BP monitoring is now required.
Sources: Lincoff et al., NEJM 2023, PMID 37326322 (TRAVERSE). Wittert et al., Lancet Diabetes Endocrinol 2021, PMID 33153698 (T4DM).
THE SCIENCE: WHY THE WARNINGS WERE WRONG, AND WHAT HRT ACTUALLY DOES
The original warnings came from the wrong study, run on the wrong women.
The WHI enrolled 16,608 postmenopausal women with a mean age of 63, averaging 12 years past their last period. Many had existing cardiovascular risk factors. The drug they received was oral conjugated equine estrogen (CEE) plus medroxyprogesterone acetate (MPA), a synthetic progestin not used in modern protocols. When the trial was halted early at 5.2 years, the resulting hazard ratios were reported as relative risks without the absolute risk context that would have told a different story.

Timing changes everything.
Approximately 10 years after menopause, estrogen receptors in coronary artery walls are still active. Estrogen reduces inflammation, prevents cell death in vessel walls, and slows plaque buildup. Wait longer and those receptors are progressively silenced. Introducing estrogen into that older biology does not restore the protective effect. It can destabilize existing plaques instead.
The ELITE trial (Hodis et al., NEJM 2016) tested this directly: 643 women, early vs late initiators, same drug. Women who started within 6 years of menopause showed significantly slower atherosclerosis progression (p = 0.04). Women who started 10 or more years out showed no benefit at all (p = 0.29). Same molecule. Opposite outcomes. Entirely because of timing.
What HRT actually helps with, when started on time.

Sources: Rossouw JAMA 2002; Hodis NEJM 2016; Fournier Int J Cancer 2005; HHS Fact Sheet Nov 2025; Lincoff et al., NEJM 2023, PMID 37326322 (TRAVERSE trial, 5,246 men, cardiovascular safety of TRT). Wittert et al., Lancet Diabetes Endocrinol 2021, PMID 33153698 (T4DM trial, testosterone and type 2 diabetes prevention). FDA Drug Safety Communication, February 28, 2025, fda.gov (testosterone labeling changes)
The 23 years it took to correct one headline!

Sources: JAMA 2002; PMC2180400; Sarrel PMC3780684; Hodis NEJM 2016; HHS.gov Nov 2025
IMPLICATIONS: FOR WOMEN AND FOR PROVIDERS
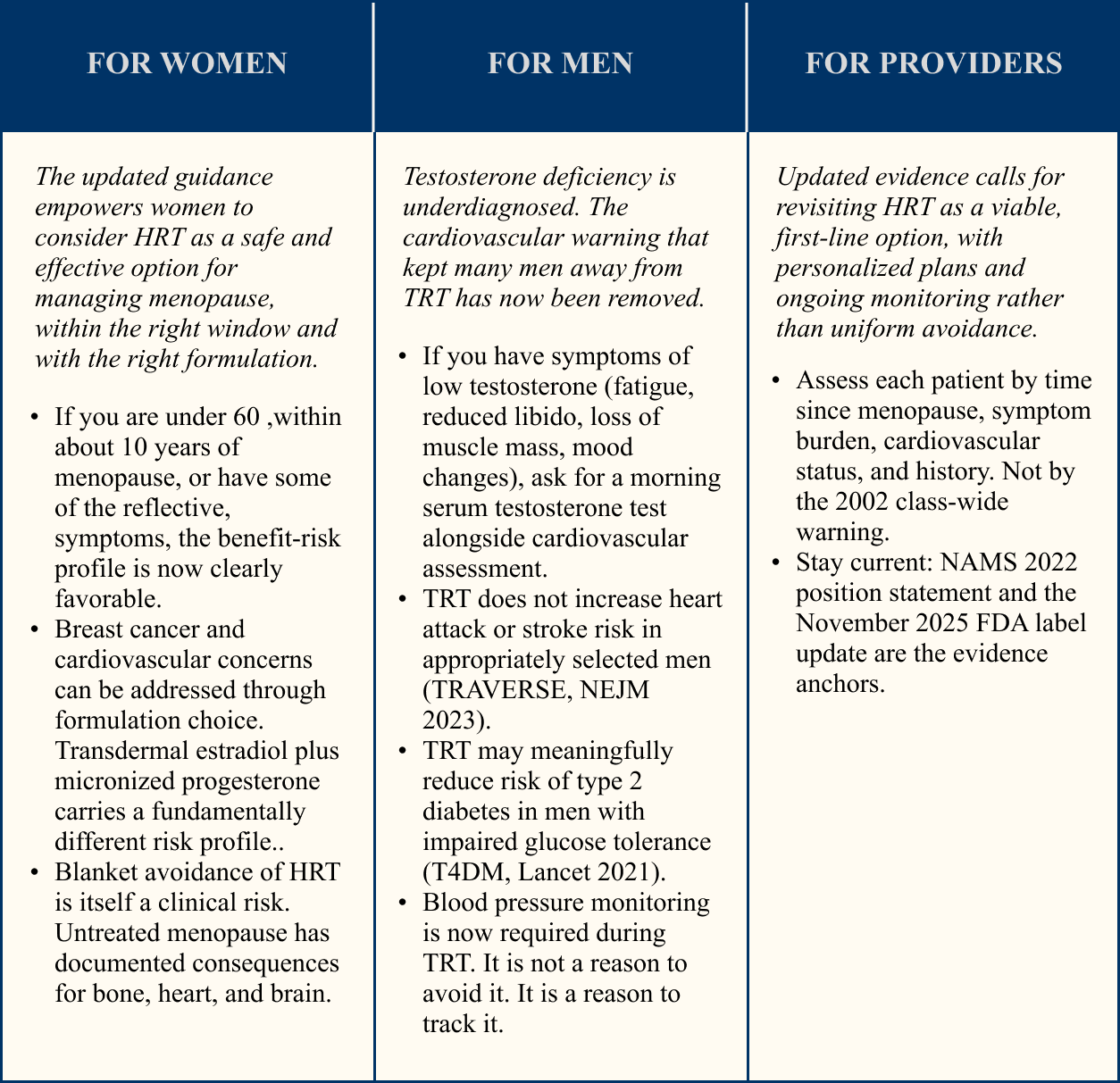
The FDA update does not make HRT right for everyone. It makes the conversation between patient and provider more important than the warning label. The implications look different on either side of that conversation.
PRACTICAL TAKEAWAYS: WHAT TO ASK AND WHAT TO DO
For women: four questions worth bringing to your next appointment.
Consulting a healthcare provider is the essential first step. These questions help you get the most out of that conversation, covering timing, dosage, formulation, and duration.
Am I around the 10-year window, or under 60? This is the most important question. The cardiovascular and cognitive benefits apply to women who initiate within about 10 years of menopause or before 60. Also, keep in mind that earlier symptoms can still also make you a candidate for therapy.
Why use an injection compared to transdermal? Injections have a more immediate action compared to transdermal absorption patches.
Do I need systemic therapy, or just local vaginal estrogen? This is dependent on other factors such as your history and current health state. Always consult with a board certified physician in scenarios such as these as there can be some minute details that a doctor will need to decipher.
For providers: what to update and where to look.
Staying current on guidelines is not optional when the evidence base has moved this significantly. Three reference points cover the key updates:
NAMS 2022 Hormone Therapy Position Statement. Publicly available at nams.org. The current clinical consensus on formulation, timing, and risk stratification.
November 2025 FDA Label Guidance. Full fact sheet at hhs.gov. Covers what changed, what remains, and the evidence review behind the decision.
ELITE Trial (Hodis et al., NEJM 2016, PMID 27028912). The only RCT designed specifically to test the timing hypothesis. 643 women, 5-year follow-up.
What ongoing monitoring looks like.

THE BOTTOM LINE
The science on HRT did not change overnight. It changed in 2005, 2013, and 2016. The FDA label changed in 2025. What matters now is that women and providers make decisions based on current evidence, not a 23-year-old headline.
If you are approaching or in menopause: bring these questions to your provider. Ask specifically about formulation and timing. The conversation is worth having.
If you are a provider: the NAMS 2022 position statement and the November 2025 FDA guidance are the two resources that cover the current evidence base most completely.
Women's health research is moving. Staying informed is the most useful thing anyone can do right now.
READER'S PULSE
This week's poll:
Your doctor brings up hormone therapy. What's your first reaction?
Sources
Rossouw et al., JAMA 2002, PMID 12117397. Fournier et al., Int J Cancer 2005, PMID 15551359 (E3N cohort, 54,548 women). Fournier et al., Breast Cancer Res Treat 2008, PMID 17333341. Hodis et al., NEJM 2016, PMID 27028912 (ELITE trial). Canonico et al., AHA Journals 2010, PMID 20601871. Sarrel et al., Am J Public Health 2013, PMC3780684. HHS Fact Sheet November 2025, hhs.gov. NAMS 2022 Position Statement, nams.org.
Nothing in this newsletter is medical advice. If you are considering hormone therapy, consult a board-certified gynaecologist or menopause specialist.

